NEOTROPICAL REGION
Etymology: Samuel T. Darling
Anopheles darlingi is regarded as the most anthropophilic of all Neotropical Anopheles, and is a highly competent malaria vector. It is easily recognized by its distinctive hindleg markings consisting of dark femora, tibia scattered with very few pale scales and Ta-III4,5 entirely white. Anopheles darlingi has one synonym—paulistensis Galvão, Lane & Correa. It is the monotypic member of the Darlingi Group and, until recently, was the only Nyssorhynchus malaria vector not yet exposed as a species complex. Substantially variant (4-5%) nuclear rDNA ITS2 sequences were reported in An. darlingi populations from Dourado, São Paulo state, in southeastern Brazil compared to typical populations.
Type locality: Caxiribu, near Porto das Caixas, Rio de Janeiro, Brazil
Type depository: U.S. National Museum, Washington, D.C., United States (USNM)
DIAGNOSTIC CHARACTERS (Click photos to view; mouse over and click large photo to zoom in.)
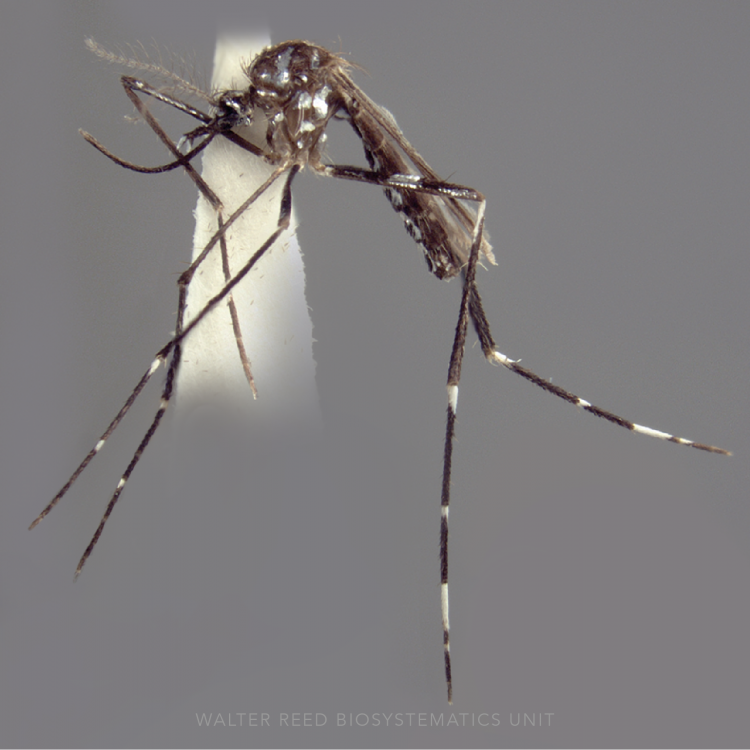
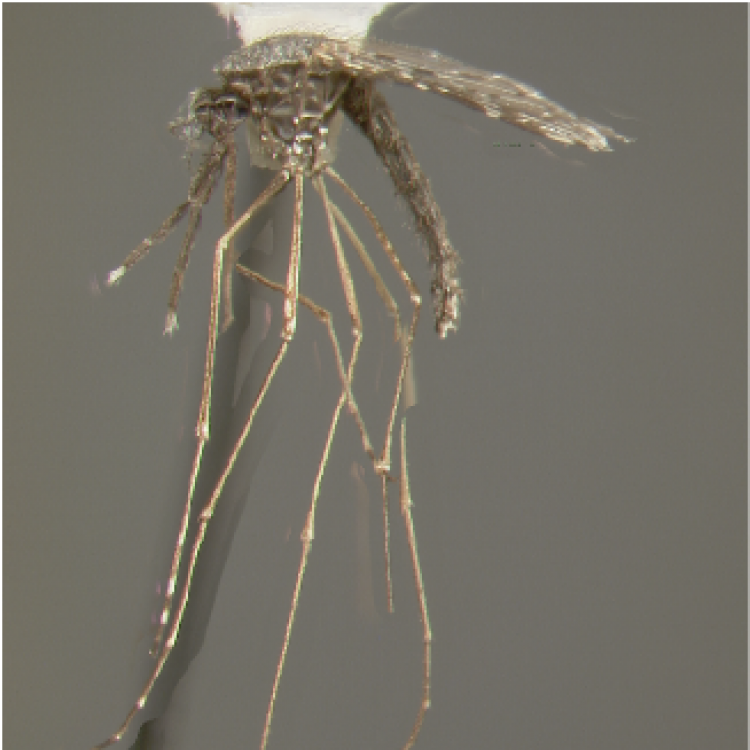
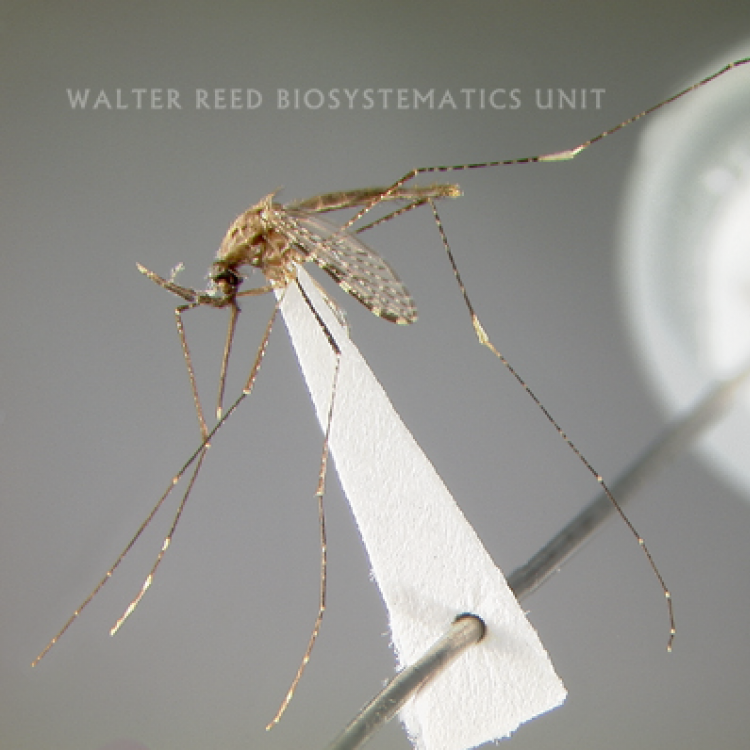
ADULT (illustrated): Head: Palpomere MPlp4 with at least some white or cream-colored scales on mediolateral surface; MPlp5 all pale. Thorax: Anterior mesepimeron with small patch of apressed scales; upper mesepimeron without pale scales. Abdomen (dorsal): Terga with posterolateral scale tufts, covered with scales; I-S bare, rarely with few, sparse scales. Wing: Accessory sector pale spot about 0.4 from wing base, conspicuously closer to sector pale than to subcostal pale spot; prehumeral dark spot well-developed, 3.0–4.0 x length of the humeral pale spot; vein 1A with fewer than five pale and five dark alternating spots. Leg: Fe-III and Ti-III dark with sparse pale scales, not speckled; Ta-III4,5 entirely white-scaled.
LARVA (not illustrated): Head: Setae 5–7-C plumose. Thorax: Seta 1-P plumose with slender branches. Abdominal segments: Seta 1 palmate leaflets smooth-sided; seta 6-IV–V single. Terminal segments: Seta 13-S well-developed, long, c. 2.2–2.5 x length of saddle.
TAXONOMIC KEYS
Pelaez 1945
Deane, L.M. et al. 1946
Causey et al. 1946
Deane, M.P. et al. 1946
Becker et al. 2010
Sallum, et al. 2020
![]()
WRBU - Genera - Central America - Adult
![]()
WRBU - Genera - Central America – Larva
![]()
WRBU - Genera - Global - Adult
![]()
WRBU - Genera - Global - Larva
![]()
WRBU - Genera - Neotropical - Adult
![]()
WRBU - Genera - Neotropical - Larva
Exemplar DNA sequences
An. darlingi COI: DQ298209–39, EU918350–62, GQ285644–55, JQ898319–27
An. darlingi whole genome: GCA_000211455.1
BIONOMICS
Immatures
Immature An. darlingi are typically found in partially-shaded, clear fresh water habitats—debris-dense stream margins, ponds with muddy bottoms, grassy ground pool and swamps in plantations or cultivated fields—always with emergent or floating vegetation. Anopheles darlingi immatures have been reportedly collected in abandoned gold mines in Venezuela and in brackish water in Belize.
Adults
Adult An. darlingi are associated with humid lowland, riverine forest-edge environment—especially in areas affected by deforestation, human settlements and agriculture—hence are closely associated with humans. They are highly anthropophilic, endophagic, but always rest outdoors. The molecularly variant Dourado population is exophilic and exophagy has been noted in sites where heavy indoor insecticidal spraying is implemented for vector control. Efforts are needed to clarify the taxonomic status of the Dourado population, especially as the only synonym—paulistensis Galvão, Lane & Correa—was described from this same location.
DISTRIBUTION NOTES
Argentina, Belize, Bolivia, Brazil, Colombia, Ecuador, El Salvador, French Guiana, Guatemala, Guyana, Honduras, Mexico, Panama, Paraguay, Peru, Suriname, Uruguay, Venezuela.

WRBU VECTOR HAZARD REPORTS
View other WRBU Vector Hazard Reports
Available GIS Models
An_darlingi_Nyari_1 South and Central America
IMPORTANT REFERENCES (full citations below)
Root 1926: 706 (M*, F*, P*, L*, E*)
Komp 1940 (distribution; Belize, Guatemala)
Komp 1942: 66 (F*), 112 (P*, L*), 152 (M*)
Rozeboom 1942a: 237 (E*; taxonomy)
Ross & Roberts 1943a: 29 (M*, F*, L*)
Pelaez 1945: 74 (F*; key)
Cova García 1946, Fig. 1 (E*), 30 (L*), 82 (F*),116 (M*)
Deane, L.M. et al. 1946 (F*, key, bionomics; NE Brazil & Amazonas)
Causey et al. 1946 (M*, key; NE Brazil & Amazonas)
Deane, M.P. et al. 1946 (L*, key, bionomics; NE Brazil & Amazonas)
Romeo Vaimonte & Castro 1951: 320 (F*)
Stone & Knight 1956b: 277 (type information)
Kreutzer et al. 1972 (chromosomes*)
Linthicum 1988: 170 (M*, F, P*, L*)
Linley 1992: 40 (E*)
Manguin et al. 1996 (L bionomics)
Lounibos et al. 1998 (molecular taxonomy)
Malafronte et al. 1999 (molecular taxonomy; s.l.)
Scarpassa & Conn 2007 (molecular taxonomy, phylogenetics)
Sinka et al. 2010: 72 (bionomics review, distribution, niche model)
Becker et al. 2010: 378 (F*, L*; key, taxonomy, distribution, bionomics)
Hiwat & Bretas 2011 (bionomics, review)
Motoki et al. 2012 (taxonomy, morphology)
Conn et al. 2013 (bionomics, distribution)
Berti et al. 2015 (distribution; Venezuela)
Emerson et al. 2015 (molecular taxonomy; s.l.; distribution)
Sallum, et al. 2020 (keys F, M, L)
CURRENT SYNONYMS
syn. paulistensis Galvão, Lane & Correa
1937: 37 (M*, F, E*; as variety). Type locality: Novo Oriente (nr. Lussanvira), Bacia do Tiete, São Paulo, Brazil (LU). References: Lane 1949 (synonymy).
CITED REFERENCES
Becker, N., Petrić, D., Zgomba, M., Boase, C., Madon, M., Dahl, C., & Kaiser, A. (2010). Mosquitoes and their control (Second ed.). Berlin Heidelberg: Springer-Verlag.
Berti, J., Guzmán, H., Estrada, Y., & Ramírez, R. (2015). New records of mosquitoes (Diptera: Culicidae) from Bolívar State in South Eastern Venezuela, with 27 new species for the state and 5 of them new in the country. Frontiers in Public Health, 2, 10.
Causey, O.R., Deane, L.M., & Deane, M.P. (1946). An illustrated key by male genitalic characteristics for the identification of thirty-four species of Anophelini from the northeast and Amazon regions of Brazil, with a note on dissection technique. American Journal of Hygiene Monograph Series, 18, 21–31.
Conn, J., Quiñones, M., & Póvoa, M.M. (2013). Phylogeography, vectors, and transmission in Latin America. In S. Manguin (Ed.), Anopheles mosquitoes-new insights into malaria vectors (pp. 145–172). Janeza Trdine 9, 51000 Rijeka, Croatia: InTech.
Cova García, P. (1946). Notas sôbre los anofelinos de Venezuela y su identificacion. (CSP12/C1). Editorial Grafolit.
Deane, L.M., Causey, O.R., & Deane, M.P. (1946). I. An illustrated key by adult female characteristics for the identification of thirty-five species of Anophelini, with notes on the malaria vectors (Diptera, Culicidae). In Studies on Brazilian anophelines from the northeast and Amazon regions (pp. 1–18). Baltimore: The Johns Hopkins Press.
Deane, M.P., Causey, O. R., & Deane, L.M. (1946). III. An illustrated key by larval characteristics for the identification of thirty-two species of Anophelini, with descriptions of two larvae. In Studies on Brazilian anophelines from the northeast and Amazon regions (pp. 33–50). Baltimore: The Johns Hopkins Press.
Emerson, K.J., Conn, J.E., Bergo, E.S., Randel, M.A., & Sallum, M.A.M. (2015). Brazilian Anopheles darlingi Root (Diptera: Culicidae) clusters by major biogeographical region. PLoS ONE, 10(7), e0130773.
Galvão, A.L.A., Lane, J., & Correa, R. (1937). Notas sôbre os Nyssorhynchus de São Paulo. V. Sôbreos Nyssorhynchus de Novo Oriente. Rev. Biol. Hyg., 8, 37-45.
Hay, S.I., Sinka, M.E., Okara, R.M., Kabaria, C.W., Mbithi, P.M., Tago, C.C., …Godfray, H.C.J. (2010). Developing global maps of the dominant Anopheles vectors of human malaria. PLoS Medicine, 7(2): e1000209.
Hiwat, H., & Bretas, G. (2011). Ecology of Anopheles darlingi Root with respect to vector importance: a review. Parasites and Vectors, 4, 177–189.
Komp, W.H.W. (1940). The occurrence of Anopheles Root in British Honduras and Guatemala. Public Health Reports, 55(16), 693–694.
Komp, W.H.W. (1942). The anopheline mosquitoes of the Caribbean Region. National Institute of Health Bulletin No. 179.
Kreutzer, R.D., Kitzmiller, J.B., & Ferreira, E. (1972). Inversion polymorphism in the salivary gland chromosomes of Anopheles darlingi Root. Mosquito News, 32, 555–565.
Lane, J. (1949). Anophelines of the Neotropical Region, pp. 399-418. In: M.F. Boyd (ed). Malariology. W.B. Saunders, Philadelphia.
Linley, J.R. (1992). The eggs of Anopheles atropos and Anopheles darlingi (Dipteral: Culicidae). Mosquito Systematics, 24(1), 40–50.
Linthicum, K.J. (1988). A revision of the Argyritarsis Section of the subgenus Nyssorhynchus of Anopheles (Diptera: Culicidae). Mosquito Systematics, 20, 98–271.
Lounibos, L.P., Wilkerson, R.C., Conn, J.E., Hribar, L.J., Fritz, G.N., & Danoff-Burg, J.A. (1998). Morphological, molecular, and chromosomal discrimination of cryptic Anopheles (Nyssorhynchus) (Diptera: Culicidae) from South America. Journal of Medical Entomology, 35(5), 830–838.
Malafronte, R. S., Marrelli, M. T., & Marinotti, O. (1999). Analysis of ITS2 DNA Sequences from Brazilian Anopheles darlingi (Diptera: Culicidae). Journal of Medical Entomology, 36(5), 631–634.
Manguin, S., Roberts, D.R., Andre, R.G., Rejmankova, E., & Hakre, S. (1996). Characterization of Anopheles darlingi (Diptera: Culicidae) larval habitats in Belize, Central America. Journal of Medical Entomology, 33(2), 205–211.
Motoki, M.T., Suesdek, L., Bergo, E.S., & Sallum, M.A.M. (2012). Wing geometry of Anopheles darlingi Root (Diptera: Culicidae) in five major Brazilian ecoregions. Infection Genetics and Evolution, 12(6), 1246–1252.
Pelaez, D. (1945). Anofelinos de México. Clave para la determinación de especies y subespecies basada en los caracteres de las hembras adultas. Ciencia, 6, 69–77.
Ponce, P., Cevallos, V., & Carrazco-Montalvo, A. (2021). Mosquitoes (Diptera: Culicidae) of Ecuador: A revised checklist, new records and species of medical importance. bioRxiv.
Romeo Viamonte, J.M., & Castro, M. (1951). Estudio de la morfología de la armadura faringea de algunos anofilinos. (Dipt. Culic.). Revista de Sanidad e Higiene Pública, 25, 313–330.
Root, F.M. (1926). Studies on Brazilian mosquitoes. I. The anophelines of the Nyssorhynchus group. American Journal of Hygiene, 6(5), 684–717.
Ross, E.S., & Roberts, H.R. (1943a). Mosquito atlas. Part I. The Nearctic Anopheles, important malaria vectors of the Americas, and Aedes aegypti, Culex quinquefasciatus. Contributions of the American Entomological Institute.
Rozeboom, L.E. (1942a). Subspecific variations among Neotropical Anopheles mosquitoes, and their importance in the transmission of malaria. American Journal of Tropical Medicine and Hygiene, 22, 235–255.
Sallum, M.A.M., Obando, R.G., Carrejo, N. et al. Identification key to the Anopheles mosquitoes of South America (Diptera: Culicidae). Parasites and Vectors, 13, 542 (2020). https://www.biomedcentral.com/collections/id-keys-anopheles
Scarpassa, V.M., & Conn, J.E. (2007). Population genetic structure of the major malaria vector Anopheles darlingi (Diptera: Culicidae) from the Brazilian Amazon, using microsatellite markers. Memórias do Instituto Oswaldo Cruz, 102(3), 319–327.
Stone, A., & Knight, K.L. (1956b). Type specimens of mosquitoes in the United States National Museum. III. The genera Anopheles and Chagasia (Diptera, Culicidae). Journal of the Washington Academy of Sciences, 46(9), 276–280.
CITE THIS PAGE
Walter Reed Biosystematics Unit (Year). Anopheles darlingi species page. Walter Reed Biosystematics Unit Website, http://wrbu.si.edu/vectorspecies/mosquitoes/an_darlingi, accessed on [date (e.g. 03 February 2020) when you last viewed the site].